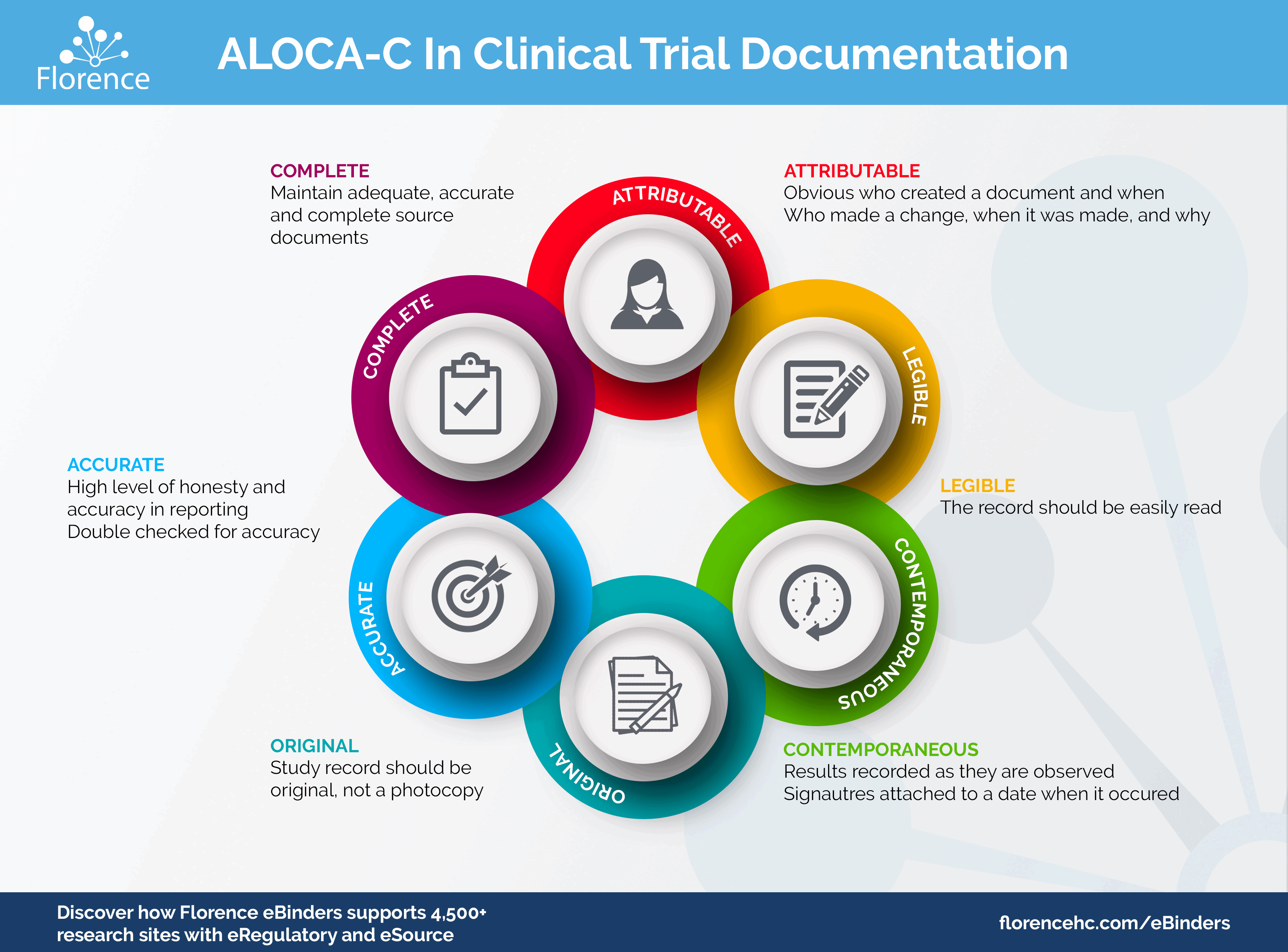
The Necessity of Clinical Research Documentation Training Programs and the Value of Learning from Mistakes - ACRP

Guidance Document: Part C, Division 5 of the Food and Drug Regulations “Drugs for Clinical Trials Involving Human Subjects” (GUI-0100) - Canada.ca

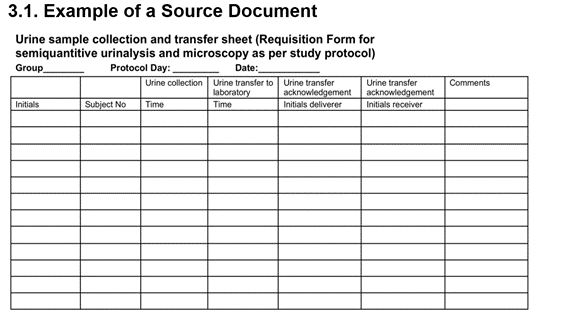
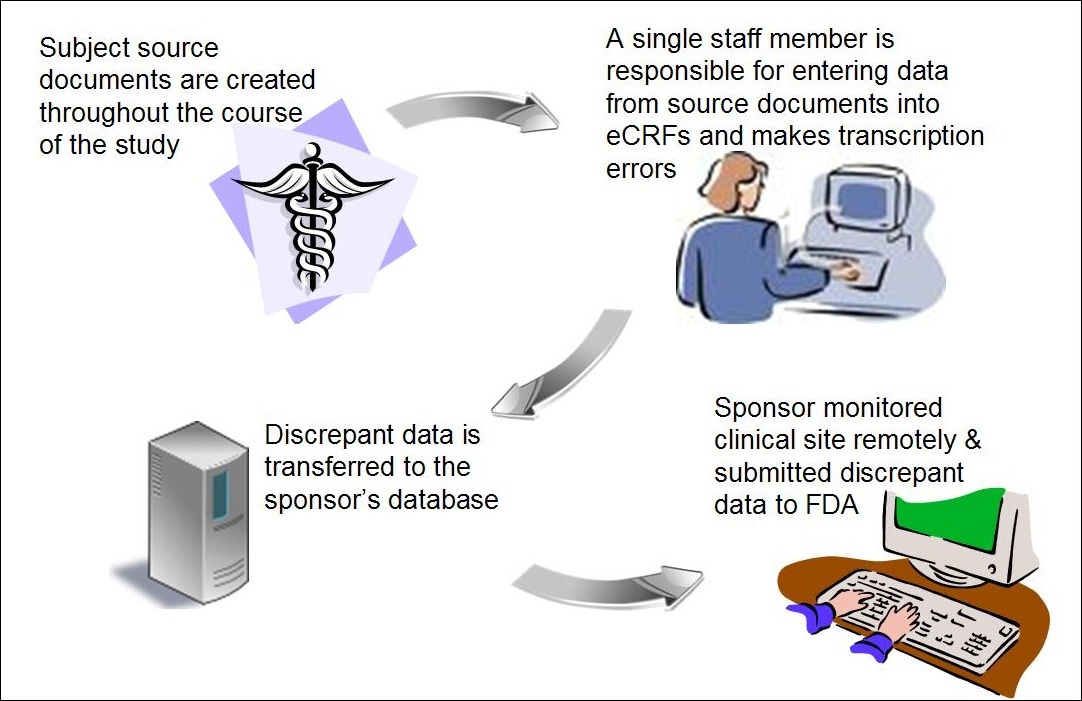
The Role of Source Data Verification (SDV) and Source Data Review (SDR) in Driving Clinical Trial Data Quality - Medidata Solutions