Enhancing pediatric clinical trial feasibility through the use of Bayesian statistics. – Pubrica Academy

Pediatric Clinical Trials Conducted in South Korea from 2006 to 2015: An Analysis of the South Korean Clinical Research Information Service, US ClinicalTrials.gov and European Clinical Trials Registries | Semantic Scholar

Pediatric Clinical Trials: Special Considerations and Requirements - Life Science Training Institute

Promoting Global Clinical Research in Children - The Multi-Regional Clinical Trials Center of Harvard and Brigham and Women's Hospital
PLOS ONE: Ten Years after the International Committee of Medical Journal Editors' Clinical Trial Registration Initiative, One Quarter of Phase 3 Pediatric Epilepsy Clinical Trials Still Remain Unpublished: A Cross Sectional Analysis

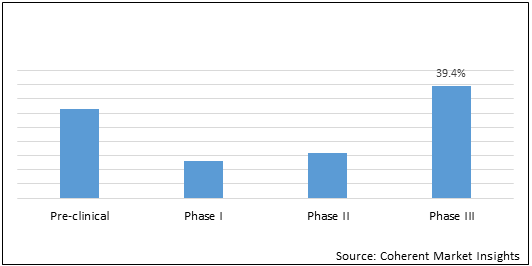
Distribution of pediatric clinical trials by classification of study... | Download Scientific Diagram
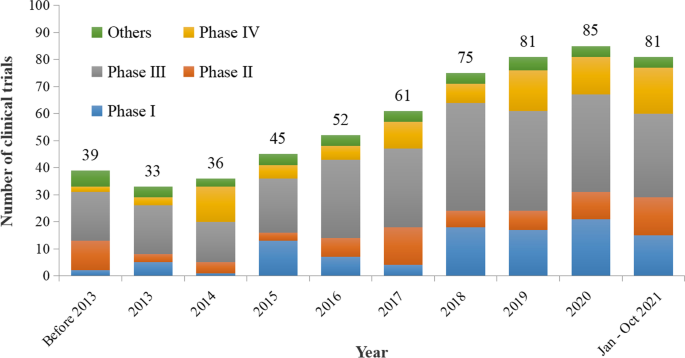
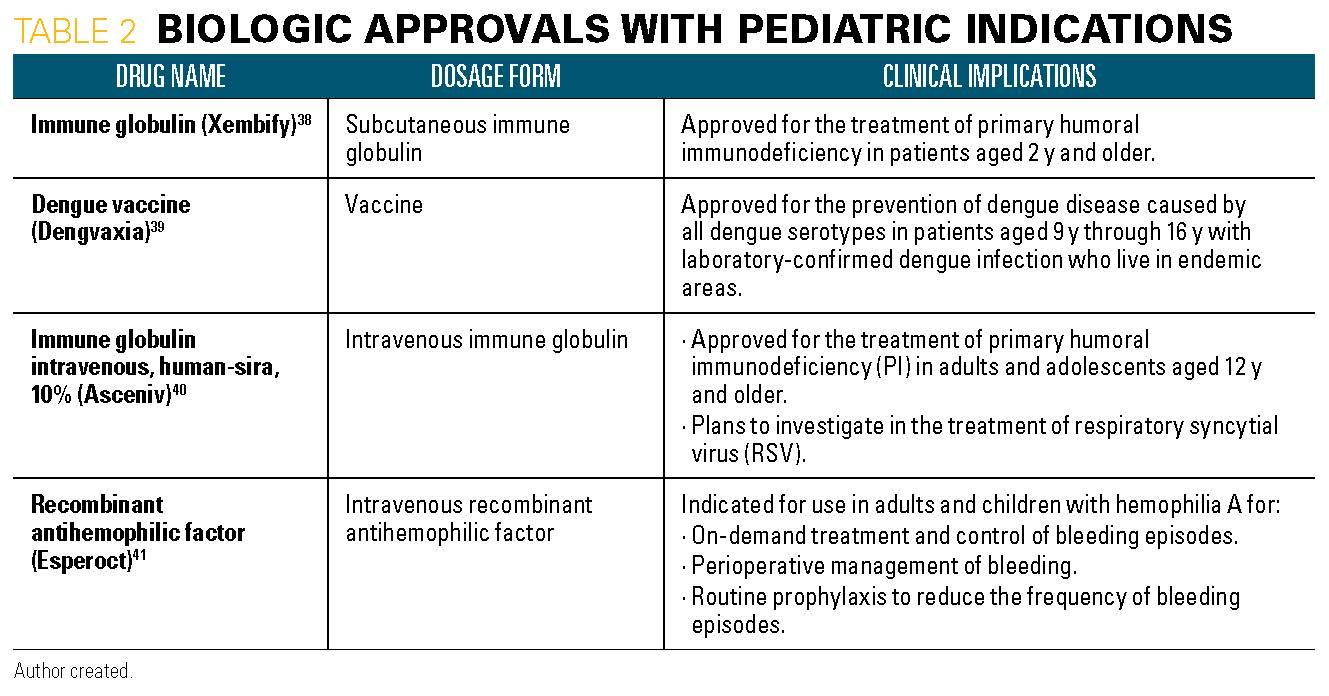
Current situation of pediatric clinical trials in China: focus on trials for drug marketing application and administrative approval | BMC Pediatrics | Full Text

1 Pediatric Clinical Trials, Lessons Learned Jennifer S. Li, MD, MHS Professor of Pediatrics and Medicine Division of Cardiology Duke University School. - ppt download

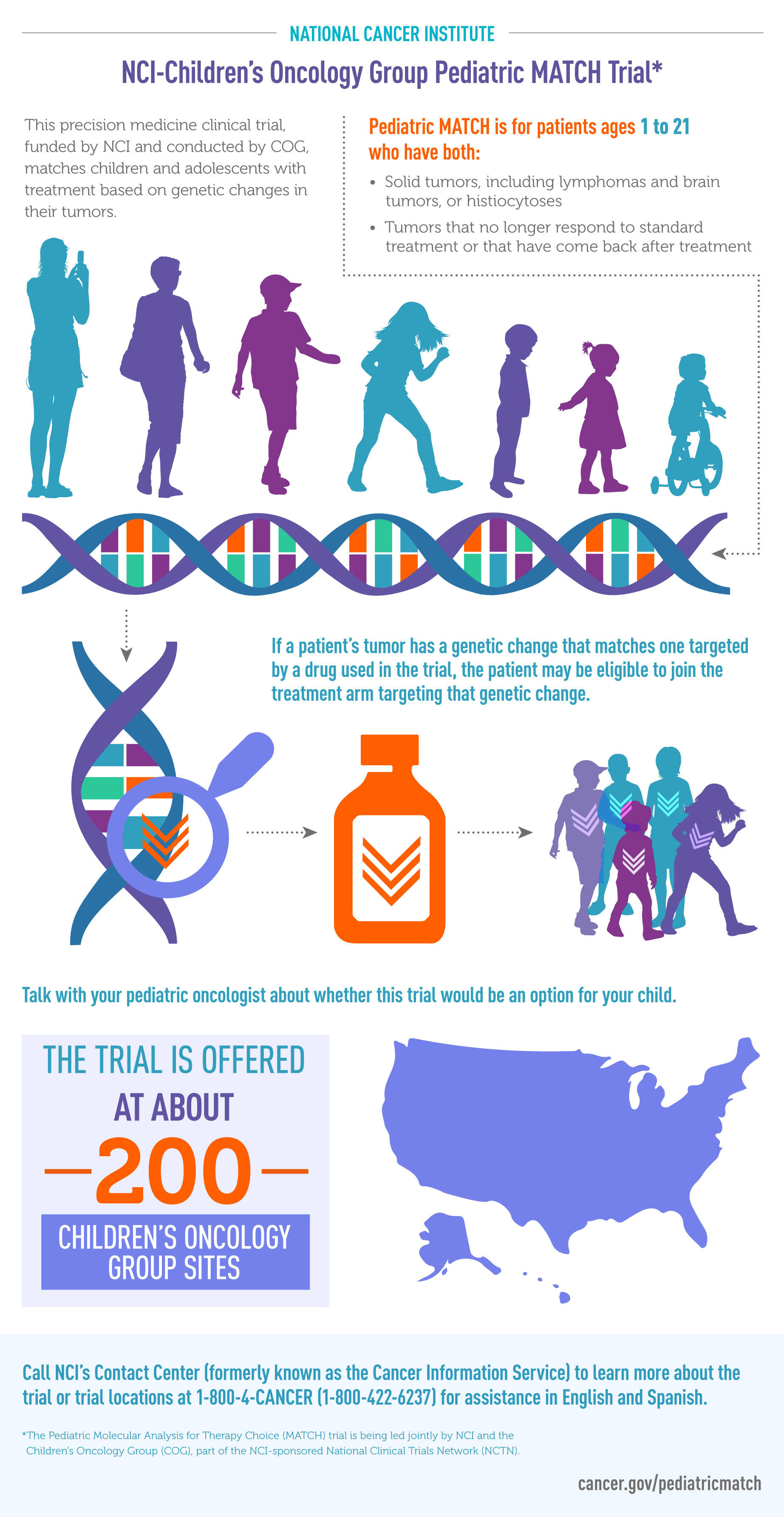
NCI-COG Pediatric MATCH trial to test targeted drugs in childhood cancers | National Institutes of Health (NIH)

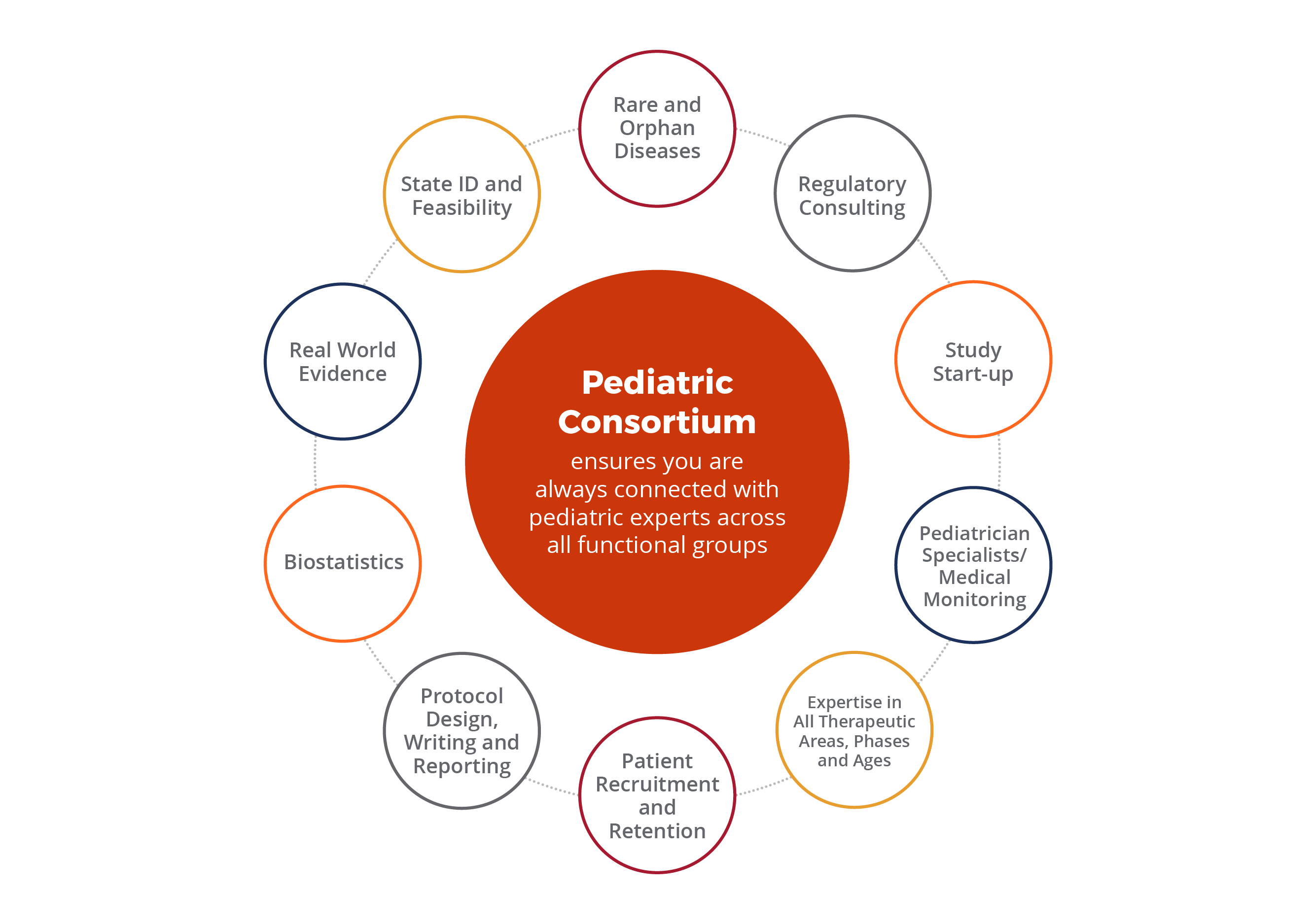
Effectively Addressing the Challenges of Pediatric Clinical Trials with Patient-Centric Best Practices | CenterWatch