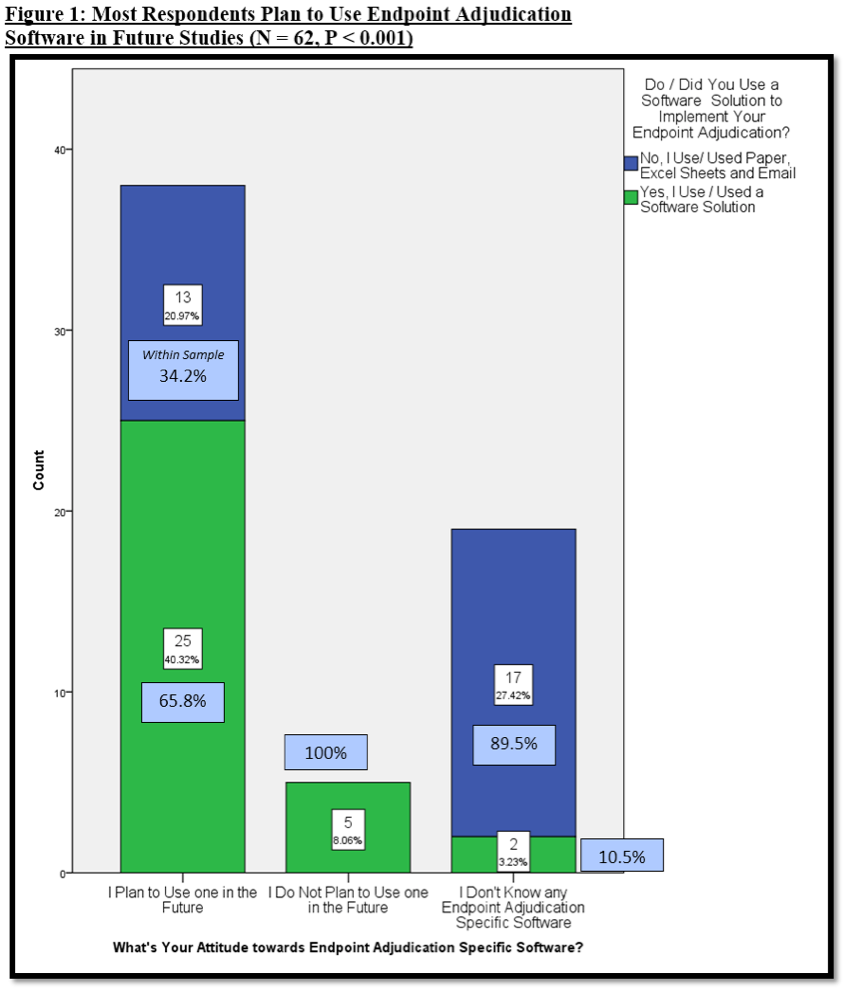
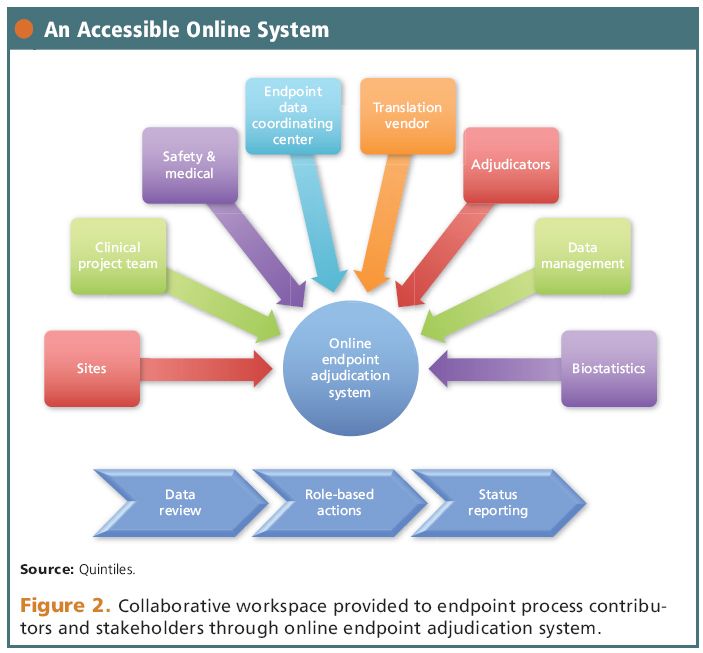
Comparative Reductions in Investigator-Reported and Adjudicated Ischemic Events in REDUCE-IT - ScienceDirect

Agreement between public register and adjudication committee outcome in a cardiovascular randomized clinical trial Erik Kjøller, MD, DMSc, Jørgen Hilden, - ppt download

7 Areas to Evaluate When Selecting Your Clinical Adjudication Partner - Paperpicks Leading Content Syndication and Distribution Platform

Guidelines for fracture healing assessments in clinical trials. Part I: Definitions and endpoint committees - Injury

Biopharmaceutical and medical device companies have made EDC (electronic data capture) a cornerstone of clinical trial and registry management because of its proven, significant process improvements over paper-based systems. EDC is a technology ...
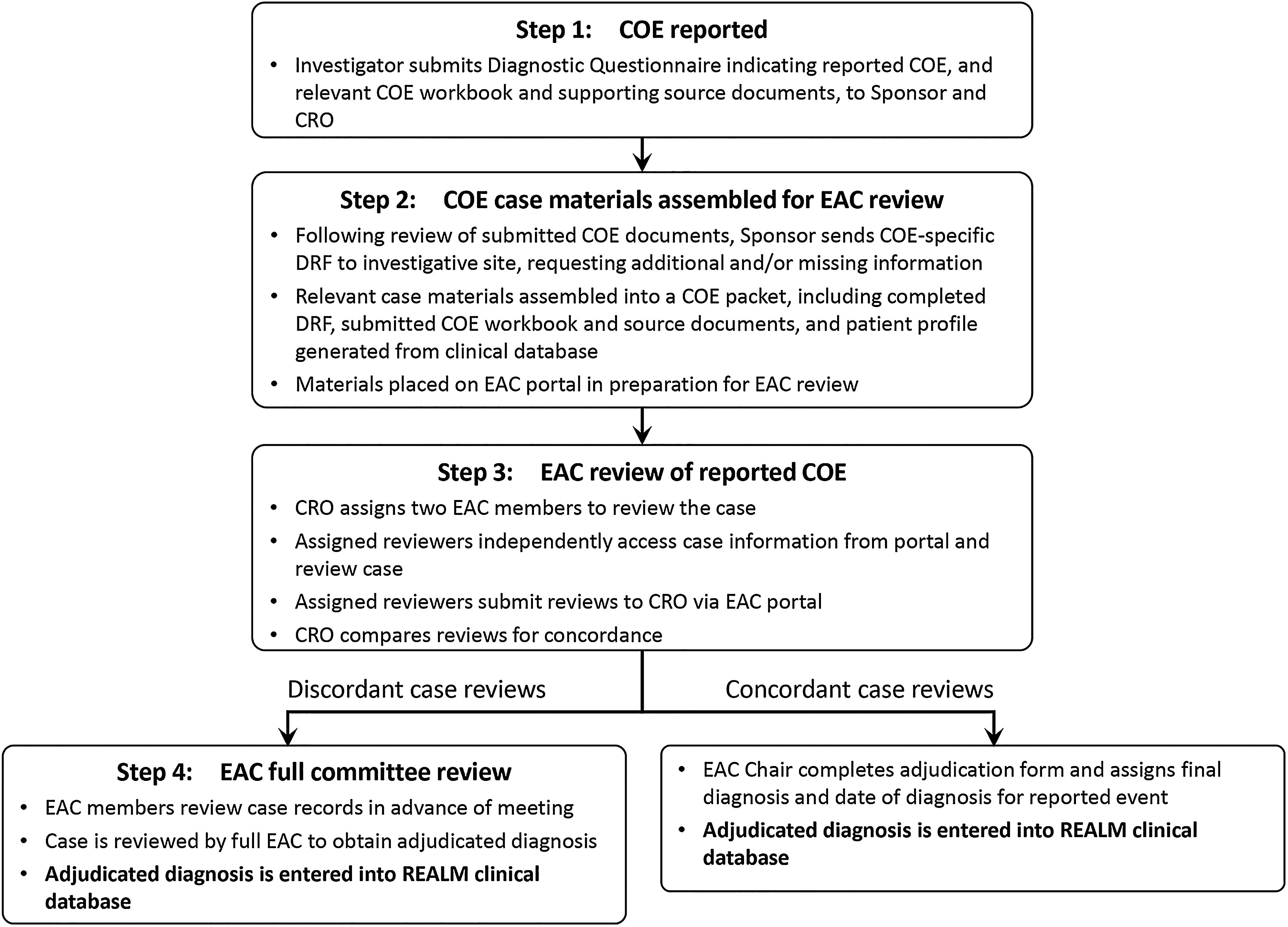
Clinical Outcome Event Adjudication in a 10-Year Prospective Study of Nucleos(t)ide Analogue Therapy for Chronic Hepatitis B
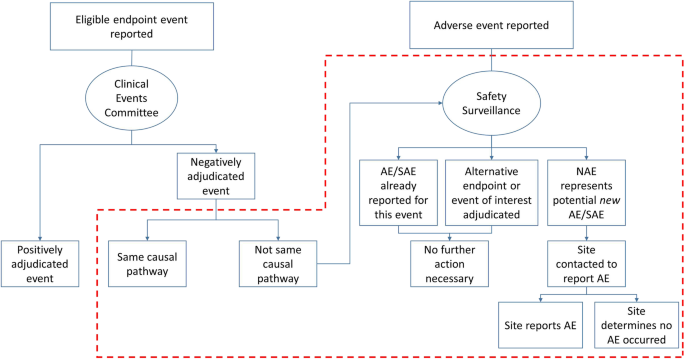
Methods for safety and endpoint ascertainment: identification of adverse events through scrutiny of negatively adjudicated events | Trials | Full Text

Central Adjudication Identified Additional and Prognostically Important Myocardial Infarctions in Patients Undergoing Percutaneous Coronary Intervention | Circulation: Cardiovascular Interventions

Clinical endpoint adjudication in a contemporary all-comers coronary stent investigation: Methodology and external validation - ScienceDirect

Determination of Hospitalization Type by Investigator Case Report Form or Adjudication Committee in a Large Heart Failure Clinical Trial (BEST) - Journal of Cardiac Failure